Glaxo SmithKline’s recent recall of Childrens’ Panadol Baby Drops has got the Hornet crew thinking about quality control even more than usual. As even the best of us make mistakes, we can all benefit from refresher quality control lessons once in a while!
The medicine itself is completely safe and compliant with all the strict requirements on pharmaceuticals. GSK are at pains to stress this on their website. Yet they still have a problem. The reason for the recall, as quoted in their press release, is ‘possible inaccurate placement of the dosing indicator on the syringe’. Check out this picture (from the Therapeutic Goods Association) and you can see how the faulty syringes could lead to a serious overdose.
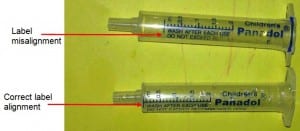 Poor Panadol. All that quality control and attention to detail in monitoring their medicine, yet they still had to recall it because an add-on product wasn’t quite right. Congratulations to GSK for taking prompt and positive action, but how did this happen in the first place?
Poor Panadol. All that quality control and attention to detail in monitoring their medicine, yet they still had to recall it because an add-on product wasn’t quite right. Congratulations to GSK for taking prompt and positive action, but how did this happen in the first place?
We’ve never had anything this serious at Hornet, but because we work with quality control day in, day out, across a huge range of products and industries, we’ve got a bit of insight.
Most likely, the problem began at the specification stage. And the fact that pharmaceuticals are so quality conscious probably made it worse. All attention was directed on the product itself, with add-ons like the syringe being examined less carefully.
It’s important to realise there’s a difference in mindset between the purchasers and the manufacturers they’re sourcing from.
The people at Panadol and GSK are thinking about a medical dosing device. To them, it’s obvious that the first line has to be positioned to indicate at a specific point, to indicate a specific amount of liquid has been drawn up into the syringe. It goes without saying. Literally.
The manufacturers are focused on the most cost-effective way to overprint lots of little plastic tubes. Getting the first line close to the narrow tip leaves little margin for error, so unless the specification actually says that’s where it needs to be, why not move it further away and make the job easier? From their perspective, it’s a simple, practical decision. Unfortunately, not the one Panadol are expecting.
Lesson Number One – the product specification matters! Detail everything. Don’t take anything for granted!
Another question to ask is how this didn’t get picked up before the syringes were distributed. It’s possible, but unlikely, that there was no quality control in place. More likely is that there were quality control measures, including at least some samples and a pre-shipment inspection, but that these processes did not pick up a problem. Why not?
None of us at Hornet know, but it wouldn’t surprise us if samples were provided and the dose indicator was printed in the correct place on them. Remember, samples are like a job interview or a first date. There’s not a conscious attempt to deceive, but there’s a natural tendency to present yourself in the best possible light you can. So the manufacturer would naturally take extra care with production and choose the very best samples available to present. Maybe they did two or three almost manually, so aligning the indicator wasn’t a major issue. Maybe they did a run of fifty or so and chose the ones which looked neatest, or most like a picture which had been supplied. So everyone was happy at sample stage.
Lesson Number Two – just because the sample looks good, don’t assume the entire production run will do. The production process for a sample is not necessarily the same as it is for a production run, and it’s human nature to take extra care during a sales process.
What about quality inspections during manufacture or before shipment? There are two possibilities here.
- inspecting every piece for quality isn’t practical. Inspection works by checking some of the product and assuming the range of variation in the total production run is the same. It’s possible that Panadol just got very unlucky, so that every item they inspected was OK but some of the ones they didn’t were misaligned.
- more likely, this is related to inadequate specification. If the position of the dose indicator wasn’t specified upfront, it almost certainly didn’t make it to the inspection checklist. So even if 100% of the finished product was inspected, it wouldn’t be marked as a fault.
Lesson Number Three – make sure your inspection checklist is as extensive and detailed as possible. If you can catch a problem before goods are packaged, shipped and distributed, it is far better in terms of time, money and company reputation.
Some More Examples
In case the syringe issue isn’t immediately relevant to you and your business, here’s a few other examples of quality control issues which Hornet have experienced and which we keep an eye out for with our clients:
- Branding issues. Be sure you specify colours precisely. Supply appropriate artwork. If the logo placement is important to you, put that in writing.
- A native speaker won’t put the label on upside down or inside out, but workers in Chinese factories are not hired for their English language ability. You may need to check this.
- If you need specific packaging to ensure items don’t get damaged in transit, specify it.
- If your product ships in parts for self-assembly at home and you don’t want the parts to rattle as if it’s broken when someone picks up the package in-store, specify that they need to be taped or wrapped.
- don’t take any functionality for granted in writing the specification. If your umbrella needs a stop so it won’t turn inside out when you open it vigorously, say so!
- If you’re ordering inflatable products, blow them up and check for leaks as part of your inspection process.
Hornet are always willing to work with you on quality control. Our experience means we often think of things you wouldn’t dream of – on the other hand, you know your product, your market and what’s important far better than we do! Above all, two heads are better than one.
If you have any other tips or examples of things slipping past quality control, we’d love to hear them! Simply drop us a line and we can add points to the list to help everyone.
